Vitamin C has been big news in the world of sepsis since the publication of a study nearly 3 years ago showing that a cocktail of high-dose IV vitamin C, thiamine and hydrocortisone was highly effective at reducing mortality in patients with sepsis. The study, conducted by a team from Eastern Virginia Medical School led by Dr. Paul Marik, showed a mortality reduction from 40.4% to 8.5% with use of the cocktail, causing a wave of media coverage in the medical and lay press, considerable controversy, and the launch of a number of studies attempting to replicate its results. ESCAVO has previously reported on Dr. Marik’s study (see here and here), as well as on related vitamin C studies, such as the recently published CITRIS-ALI.
Dr. Marik’s study has divided the critical care community, with some clinicians deriding its results as implausible and bad science, some expressing cautious optimism, but waiting for further confirmatory studies, and some embracing it and even putting the treatment to use in daily clinical practice before formal evidence from randomized trials is available.
Nearly 3 years after its publication, and with confirmatory results from randomized trials still pending (the VICTAS and ACTS trials with this treatment should be reporting results soon), we at ESCAVO wanted to find out how many clinicians are actually using this treatment and what they are finding. So between September 27th and October 3rd 2019 we surveyed users of our Sepsis Clinical Guide app on their use of Dr. Marik’s vitamin C cocktail in clinical practice. This article reports the results of our survey, which, as far as we know, is the largest to date on use of this treatment in everyday practice.
Survey Results
A total of 668 users of our Sepsis Clinical Guide app from 88 countries responded to our survey, the majority of whom were physicians in critical care, emergency medicine and other specialties. As with previous ESCAVO surveys, this survey was conducted in the English and Spanish languages, with 430 respondents in the English survey representing North America, Europe, India, Africa, Middle East, Asia and Brazil, and 238 in the Spanish survey representing primarily Latin America (except Brazil) and Spain.
This section lists the survey’s questions and responses, in total and broken down by individual language survey. Questions 4, 5, 6 and 7 were only shown to respondents who indicated using the cocktail in clinical practice (answered Yes to question 3), whereas question 8 was only shown to respondents who indicated not using the cocktail (answered No to question 3).
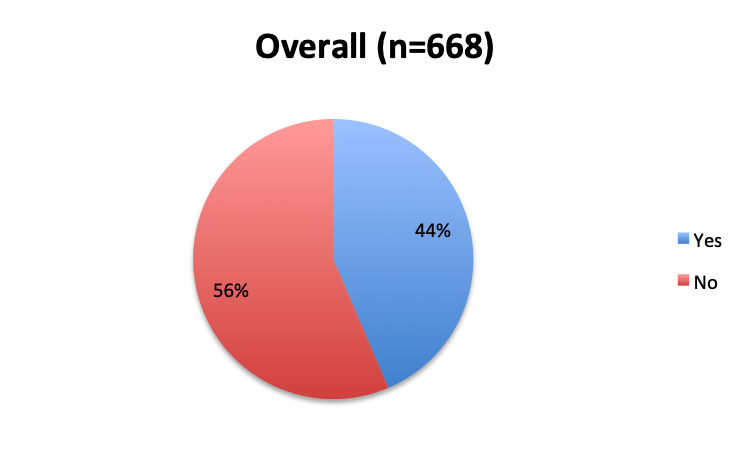
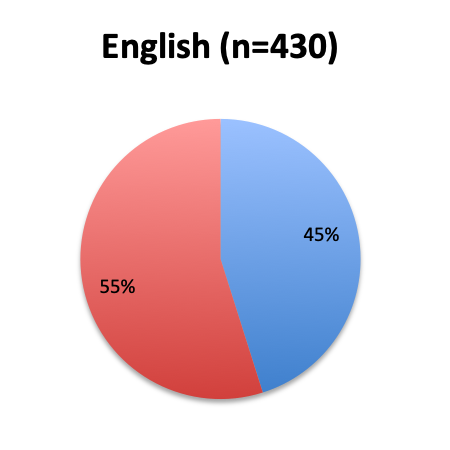
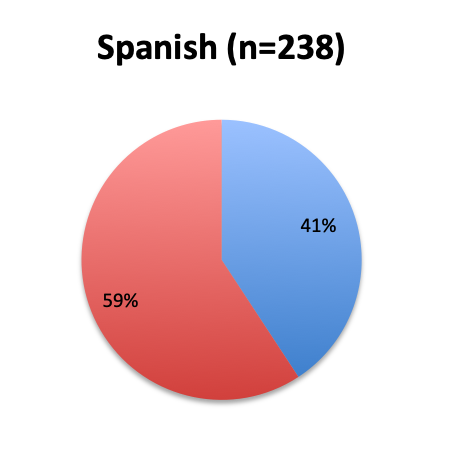
Question 1. Are you aware of Dr. Paul Marik’s study in CHEST and the Vitamin C protocol tested in the study?
A. Yes
B. No
 |
|
 |
 |
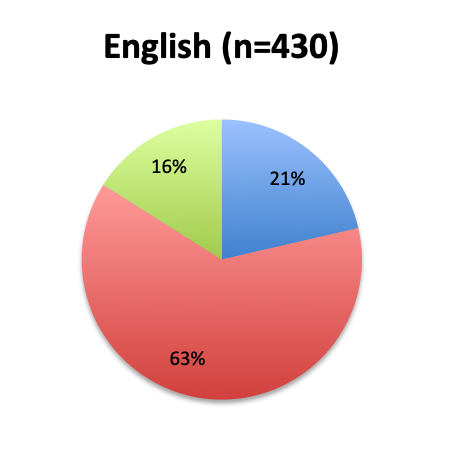
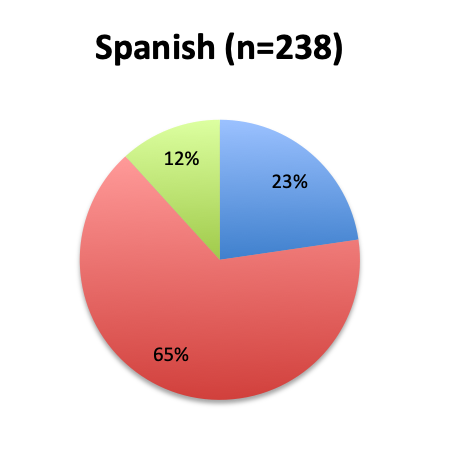
Question 2. Is this Vitamin C protocol being used to treat septic patients at your institution?
A. Yes
B. No
 |
|
 |
 |
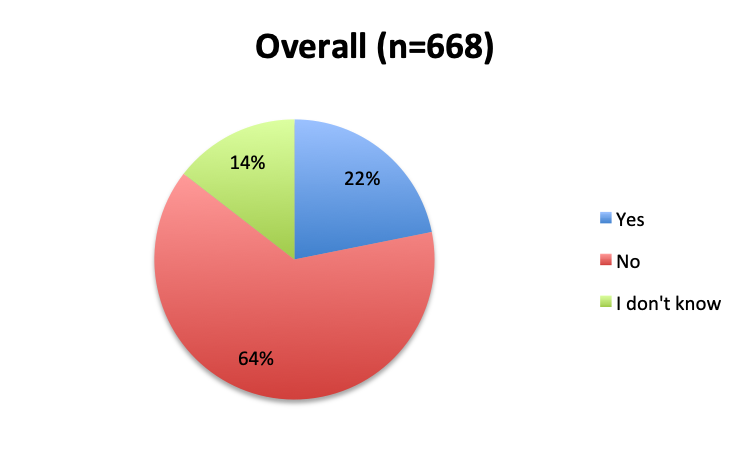
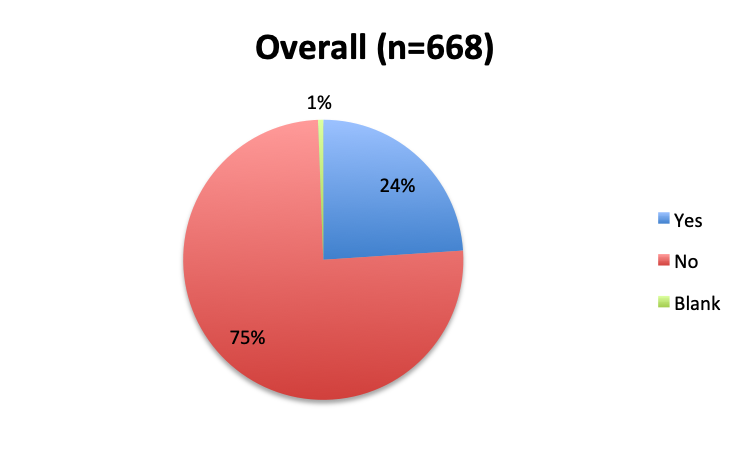
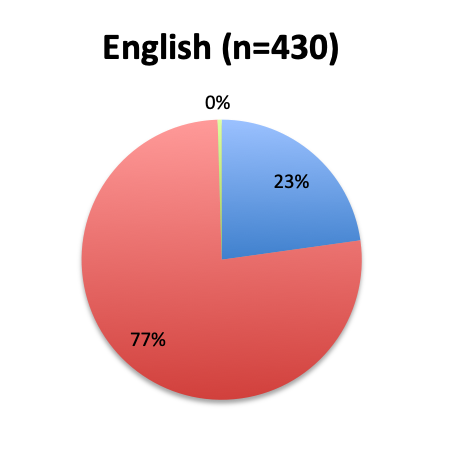
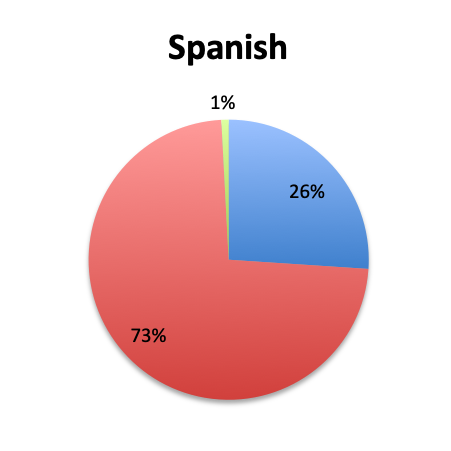
Question 3. Have you used the vitamin C protocol to treat septic patients in your care?
A. Yes
B. No
 |
|
 |
 |
Questions 4 through 7 were only asked of respondents who indicated using the vitamin C protocol in question 3
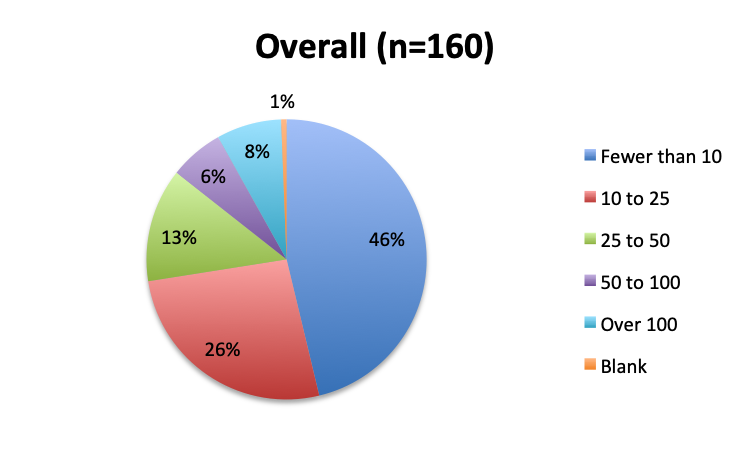
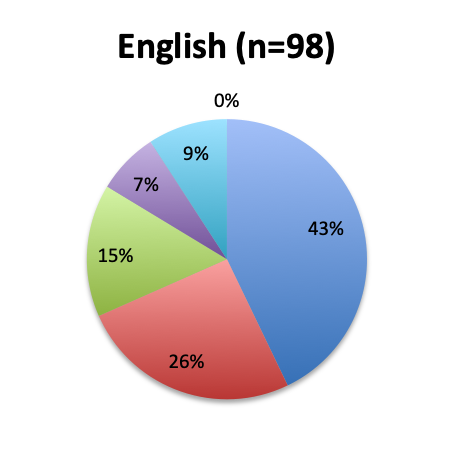
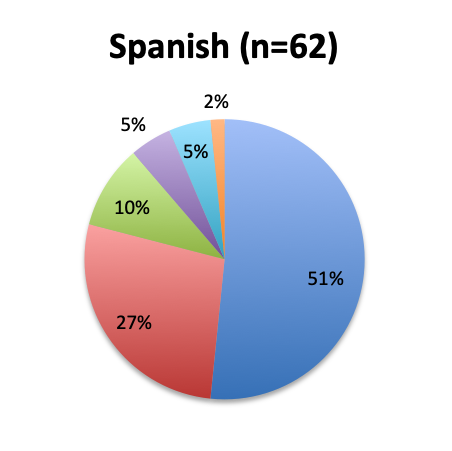
Question 4. Approximately how many sepsis patients have you treated with the vitamin C protocol?
A. Fewer than 10
B. 10 to 25
C. 25 to 50
D. 50 to 100
E. Over 100
 |
|
 |
 |
Question 5. When and in what types of septic patients is the vitamin C cocktail administered?
A. The cocktail is administered in all septic patients as soon as a diagnosis of sepsis is made
B. Only patients with severe sepsis and septic shock receive the cocktail
C. Only patients in whom all traditional sepsis treatment methods have failed receive the cocktail as a last resort
D. It varies / I don’t track this / not applicable
 |
|
 |
 |
Question 6. How has use of the Vitamin C protocol impacted mortality in your septic patients?
A. No significant difference in mortality
B. <10% mortality reduction
C. 10-30% mortality reduction
D. 30-50% mortality reduction
E. >50% mortality reduction
F. I have not tracked mortality and cannot say
 |
|
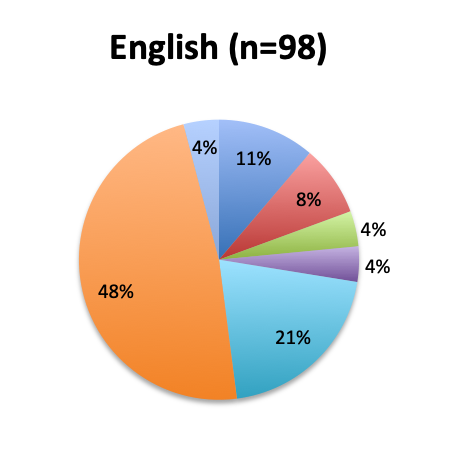 |
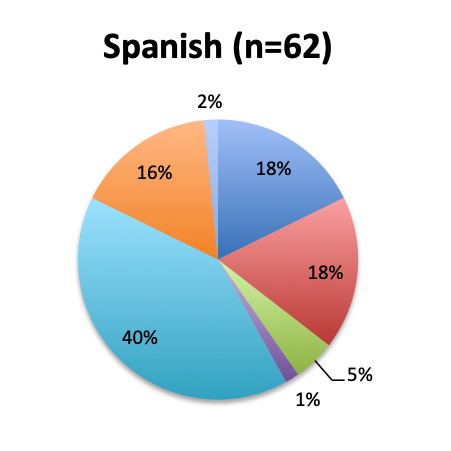 |
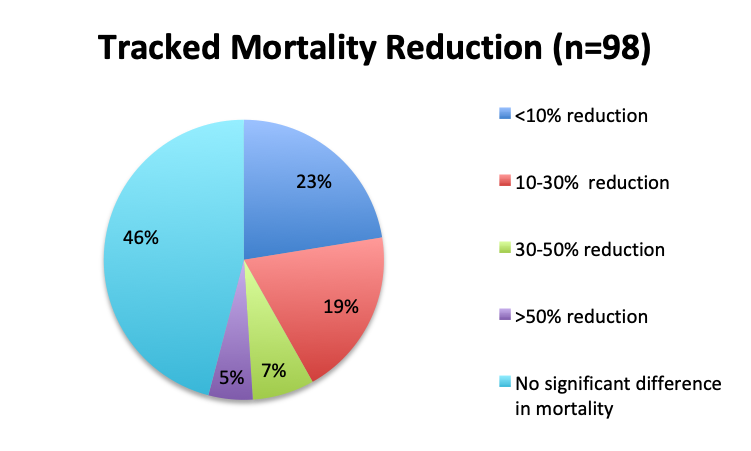 |
|
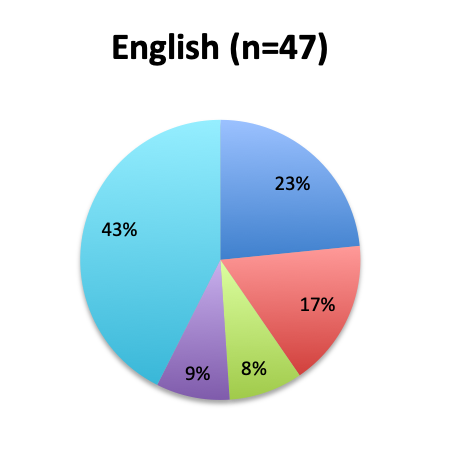 |
 |
 |
|
 |
|
| Note: These charts include all respondents who answered the mortality benefit question in both the English and Spanish surveys, but exclude respondents who left blank the patient volume and treatment timing questions. |
Question 7. Have you observed any other benefits of using vitamin C in your septic patients (select all that apply)?
A. Faster patient recovery
B. Less use of vasopressors or other agents
C. Less organ dysfunction
D. Fewer complications and sequelae
E. Other benefits (user free text entry)
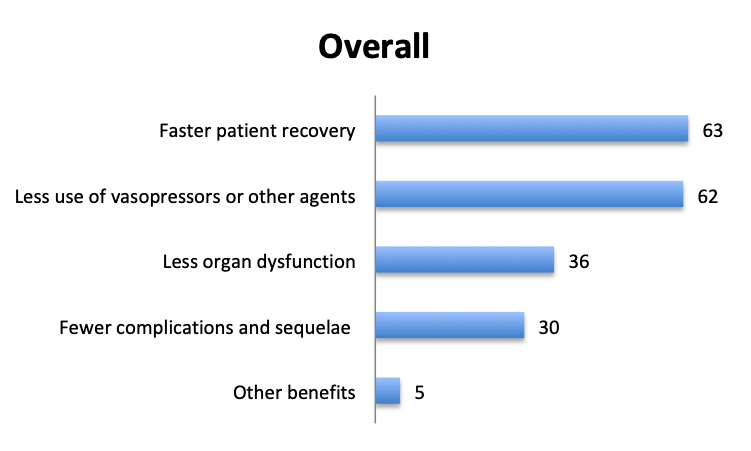 |
|
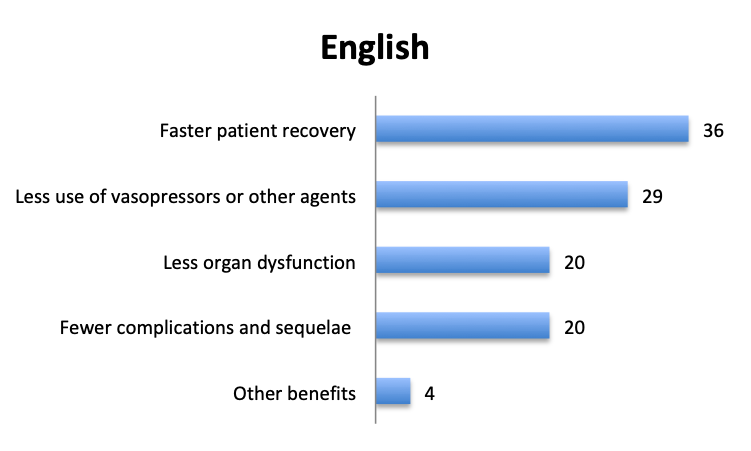 |
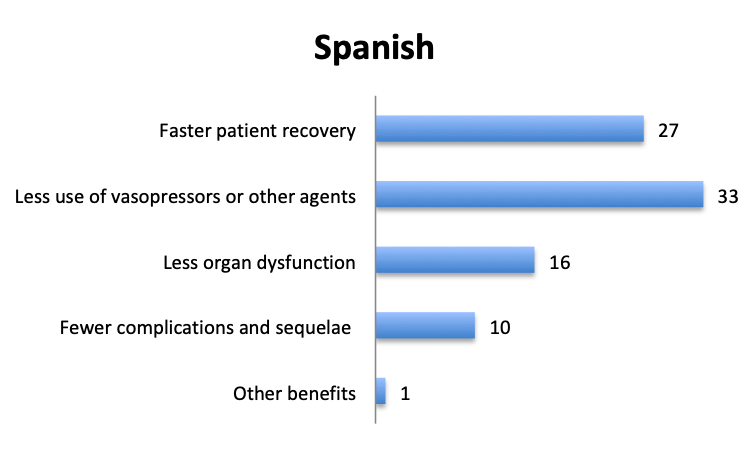 |
Question 8 was asked only of respondents who indicated not using the cocktail in question 3.
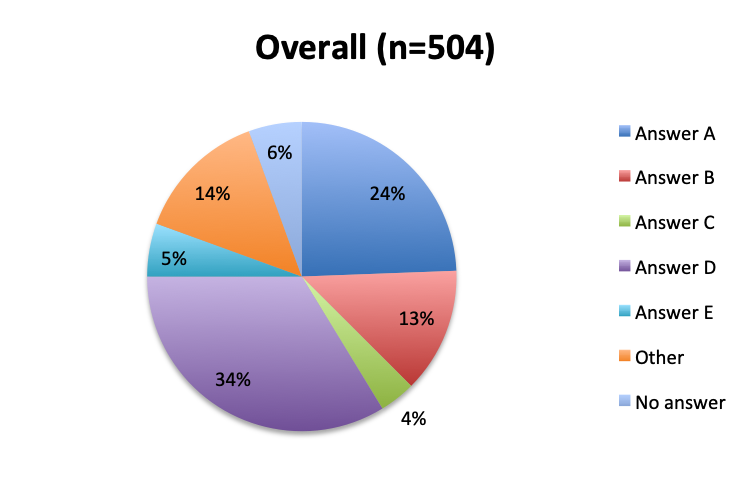
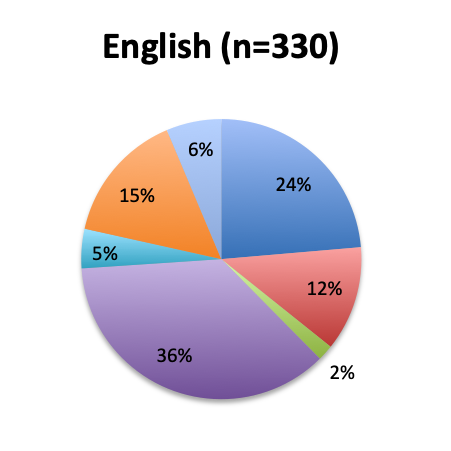
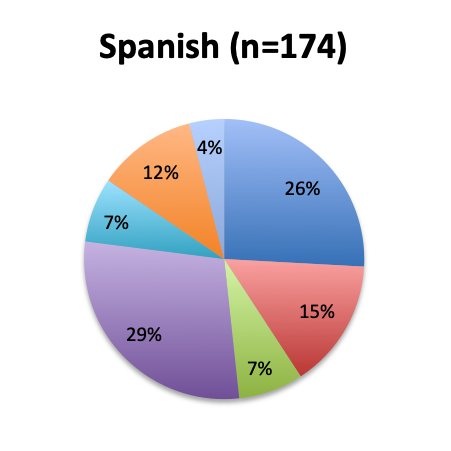
Question 8. Why have you not used the Vitamin C protocol in your septic patients?
A. I would like to try it, but my institution does not allow the use of experimental, unvalidated treatments
B. The ingredients of this cocktail are not readily available at my institution
C. I have safety concerns with this treatment regimen
D. I want to wait until Dr. Marik’s study is validated by larger randomized clinical trials before I try this treatment
E. I am very skeptical of Dr. Marik’s study and do not think this cocktail can make a significant difference in sepsis outcomes
F. Other reason (user free text entry)
 |
|
 |
 |
Question 9. Please enter any additional comments you may have about use of the Vitamin C cocktail in the treatment of sepsis. (user free text entry)
| English Survey Free Text Comments | |
|
|
| Spanish Survey Free Text Comments | |
|
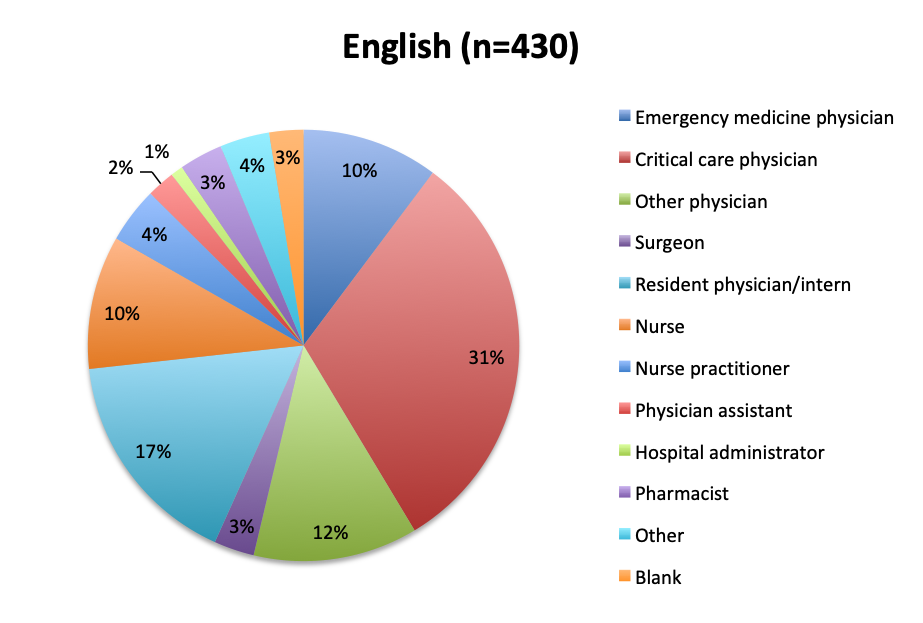
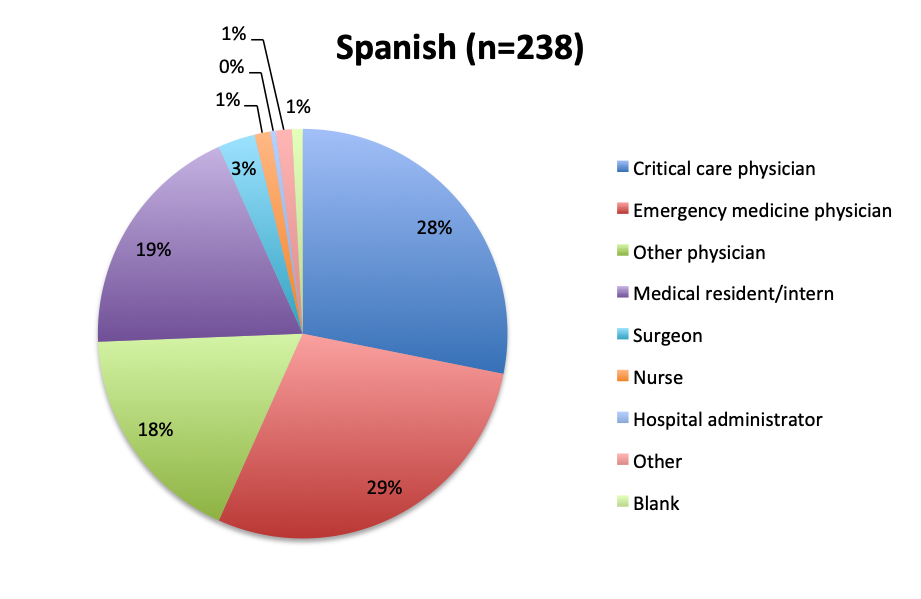
Question 10. Please select the choice that best describes your role.
 |
|
 |
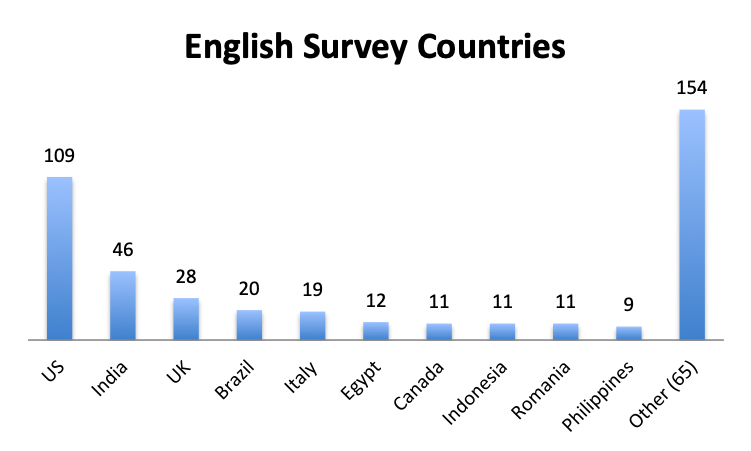
Participating Countries
 |
|
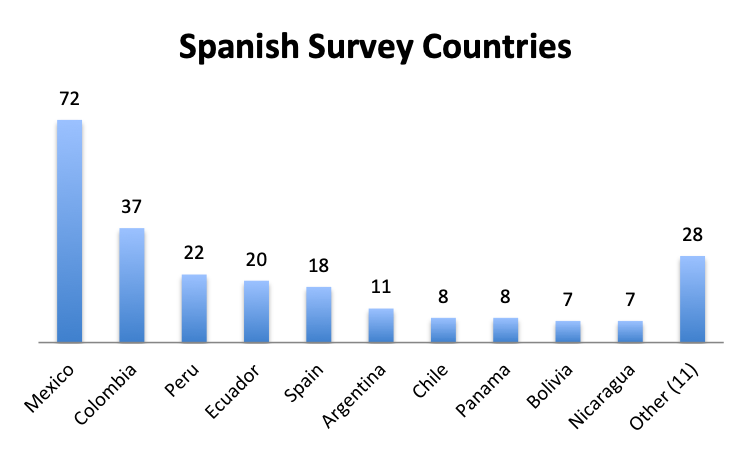 |
Results Analysis
Of the 668 total survey respondents, 160 (24%) reported using the Marik vitamin C cocktail in their treatment of septic patients. These results were consistent across different geographies, with 23% indicating use of the cocktail in the English survey and 26% in the Spanish survey. Overall, only about half the respondents (44%) reported being aware of Dr. Marik’s original study and this treatment.
Among the 160 users of the cocktail, the large majority (46%) reported using it in fewer than 10 patients, 26% reported using it in 10-25 patients, and only 12 respondents (8%) reported using the treatment in over 100 patients. Among these, 3 were from India, and 1 each from the US, Mexico, Venezuela, Nicaragua, Azerbaijan, South Africa, Turkey, Indonesia and Moldova.
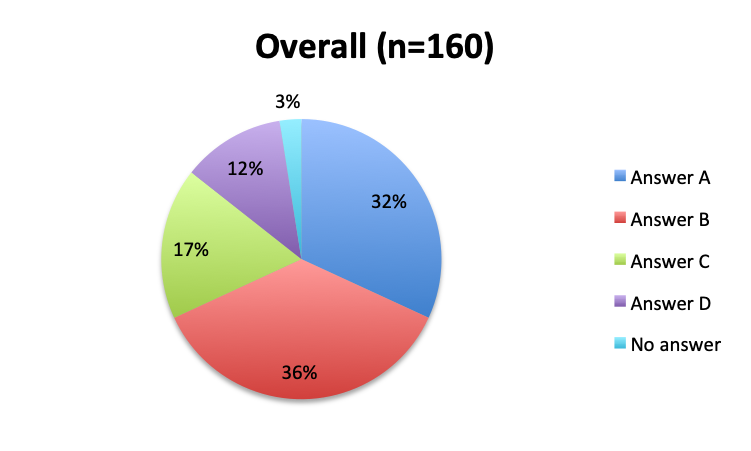
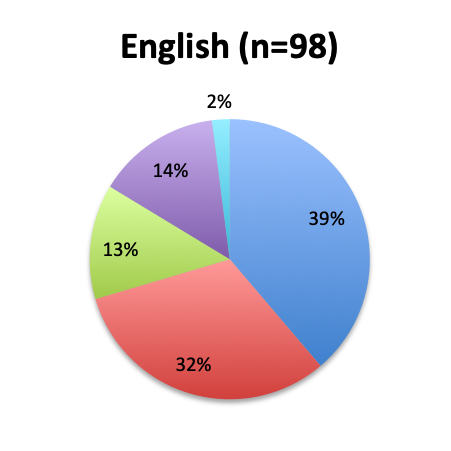
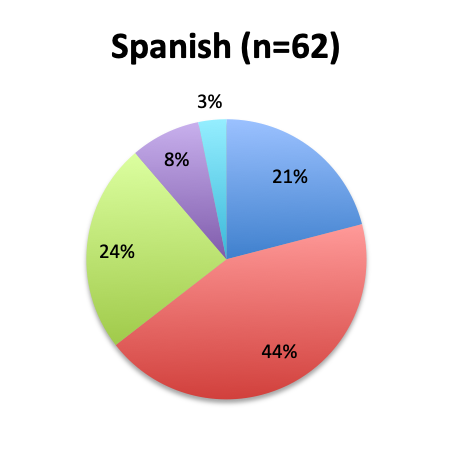
The greatest proportion of respondents (36%) reported using the vitamin C cocktail only in patients with severe sepsis and septic shock, while a slightly lower percentage (32%) reported administering it as soon as a diagnosis of sepsis is made. Only 17% of respondents indicated giving the treatment as a last resort when other treatments have failed. Interestingly, significantly more respondents in the English survey than the Spanish survey (39% vs. 21%) indicated using the cocktail in all sepsis patients as soon as diagnosis is made. In the Spanish survey, most respondents indicated using it only in patients with severe sepsis and septic shock or as a last resort, 44% and 24%, respectively. These results seem to indicate that practitioners in Latin America are on average using the treatment later in the course of the disease and in more severe cases or as last resort, whereas those in other parts of the world are using it earlier in the course of the disease.
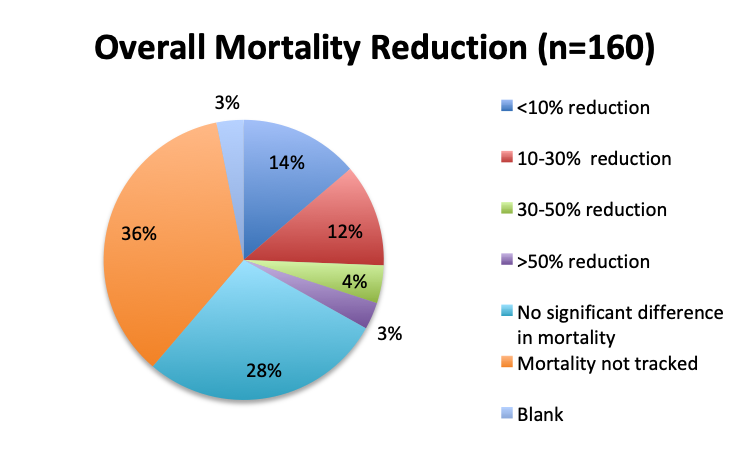
We next looked at perhaps the most important question of this survey, namely, the observed mortality benefit. Among the 160 treatment users, only 98 respondents (61%) reported tracking mortality while 36% reported not tracking mortality or left the question blank (3%). Among the 98 respondents who tracked mortality, only about half (54%) reported seeing some mortality reduction, while 46% reported no benefit. Among those who reported a mortality benefit, the majority (42%) reported seeing a mortality reduction of less than 30%, while relatively few reported seeing a reduction greater than 30% (12%). The reported mortality reduction was generally similar between the English and Spanish surveys, except that a higher proportion of respondents reported a >50% reduction in the English survey (9% vs. 2%).
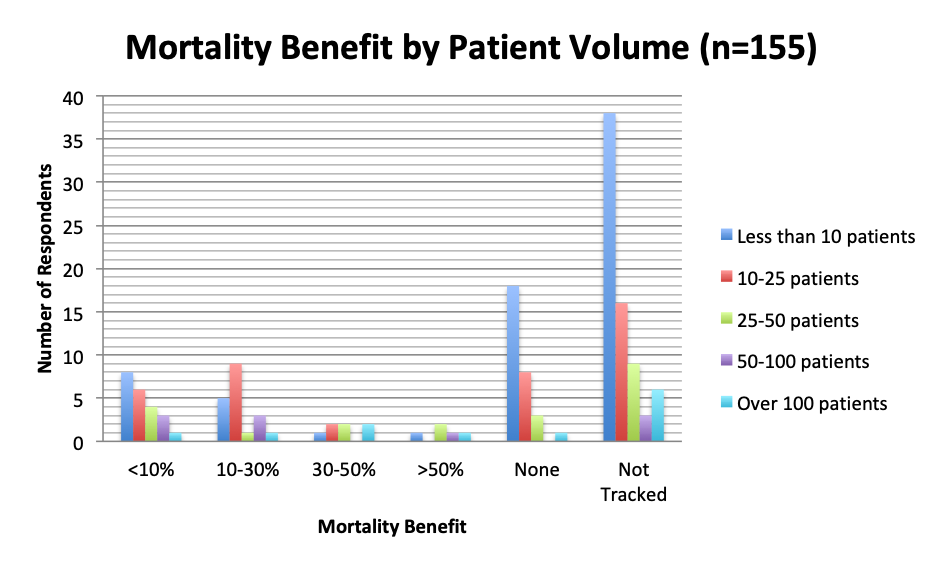
We also examined the effects of patient volume and treatment timing and patient type on mortality. In general, respondents who treated more patients tended to report a higher mortality benefit. In the 10-30% mortality reduction range, in particular, the number of respondents in the 10-25 patient group was almost double that in the <10 patient group (9 vs. 4), despite the 10-25 patient group being about half the size of the <10 patient group (42 vs. 74). This indicates that the number of 10-25 patient respondents reporting a mortality of 10-30% was almost 4 times larger than expected, suggesting a strong trend for this mortality range in this group of respondents. Additionally, most respondents who treated more than 25 patients (n=21) reported some mortality benefit distributed across all mortality reduction ranges, while only 4 respondents in these groups, including 1 in the >100 patient group, reported no benefit.
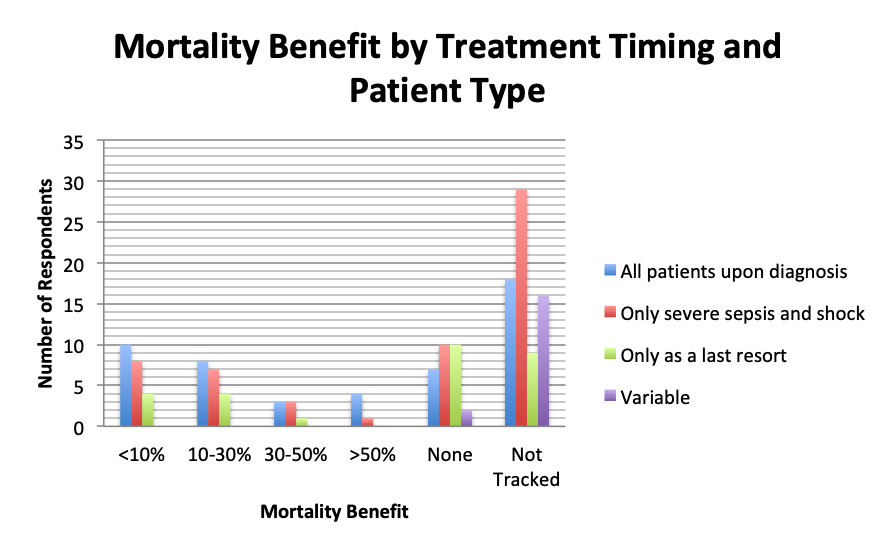
With regard to treatment timing, more respondents in the early treatment group (who treated all patients upon sepsis diagnosis) tended to show some mortality benefit than respondents who gave the cocktail only in severe sepsis and septic shock or as a last result. This effect was particularly pronounced in the >50% mortality reduction group, where 4 times as many respondents in the early treatment group than the severe sepsis treatment group reported this reduction. These results suggest earlier treatment may lead to greater mortality reduction.
In addition to mortality, we also looked at other potential benefits of the vitamin C cocktail. About 60 respondents reported benefits included “faster patient recovery” and “less use of vasopressors and other agents”, while about half that number reported “less organ dysfunction” and “fewer complications and sequelae”. The relative proportions of these responses were similar in the English and Spanish surveys, although in the Spanish survey more respondents reported less use of vasopressors.
Among the 504 respondents who reported not using the vitamin C cocktail, the majority (34%) listed the reason as a wish to wait for further validation of Dr. Marik’s study by larger randomized trials. The second most frequently given reason (24%) was that their institution does not allow use of experimental, unvalidated treatments despite their wish to try it, while 13% reported the ingredients are not readily available at their institution. Few respondents reported having an issue with the safety of the treatment (4%), and only 5% reported strong skepticism for the validity of the original vitamin C study. These results seem to indicate that most respondents who have not used the treatment are cautiously optimistic and want to wait for larger studies, or do wish to use it, but cannot do so due to institutional rules or availability reasons. Responses were fairly similar between the English and Spanish surveys, although a somewhat higher proportion of Spanish responders reported a safety concern with the treatment (7% vs. 2%).
Most user free-text comments were supportive of the treatment, cautiously optimistic, or indicated unawareness of this treatment regimen. Some of the more interesting responses included the observation that IV vitamin C adds to the fluid volume. Clearly, the volume of IV fluids given during resuscitation should be adjusted to account for the volume of fluid in the IV vitamin C bag. One respondent from Panama noted that the care team there administers vitamin C by nasogastric tube due to lack of IV formulations, which we found rather interesting.
As in all of our surveys, the majority of participants were physicians, primarily emergency medicine and critical care practitioners. In the English survey, 73% were physicians, 10% nurses, and 17% other healthcare professionals, while in the Spanish survey, an overwhelming 97% of respondents were physicians.
Over 80 countries were represented in the survey, with the biggest participants being the US, India and the UK in the English survey, and Mexico, Columbia and Peru in the Spanish survey.
Discussion
Given that the Marik vitamin C cocktail has not been validated by randomized trials or endorsed by major medical organizations, it is not surprising that the majority of survey respondents reported not using it in their clinical practice. Nonetheless, a fairly significant number, about a quarter of the total, did report using this treatment despite the lack of formal evidence. This reflects the considerable enthusiasm generated by Dr. Marik’s initial study, continued anecdotal reports of success with use of this cocktail, the relatively benign nature of its ingredients, and its fairly low cost.
Out of those who did not use the treatment, about a third reported wishing to wait for validation from larger randomized clinical trials. With the VICTAS and ACTS randomized trials recently completing enrollment, more evidence should soon be forthcoming. It was interesting to see that another third of non-users reported not administering the treatment due to external factors such as institutional policies against experimental treatments, or simply a lack of availability for the ingredients. IV vitamin C, in particular, seems to be in short supply in some areas as suggested by one of the respondents in the comments. Very few respondents refrained from using the treatment out of great skepticism about its efficacy or safety concerns. Overall, these results indicate that most clinicians are cautiously optimistic about this treatment and generally supportive of it, but waiting to see more data.
The majority of those who reported administering the cocktail not surprisingly did so in relatively few patients (fewer than 25), but surprisingly, a fair number of respondents reported treating over 50 and even over 100 patients, indicating a strong commitment to this treatment among some practitioners. Interestingly, a somewhat higher number of respondents reported administering the vitamin C cocktail only in patients with severe sepsis and septic shock and not in all sepsis patients on initial diagnosis. In the original Marik paper, the cocktail was also administered in patients with signs of “severe sepsis” and higher risk of organ dysfunction using a procalcitonin cut-off >2 ng/ml, so this may be interpreted as following the methodology of the original paper. However, the current consensus is that the cocktail should be given earlier in the course of the disease as soon as the sepsis is diagnosed in order to halt the metabolic disturbance and the organ damage it might cause sooner rather than later. Respondents in the Spanish survey, in particular, seemed to prefer administering the treatment later in more severe cases or as a last resort, perhaps indicating greater hesitancy with its use. The ever-shifting definition of sepsis is also a factor that may also potentially be muddling the waters here. What used to be defined as “severe sepsis” is now defined as just “sepsis” under the Sepsis-3 definitions.
In terms of mortality benefits, it would appear that the large benefits reported in Dr. Marik’s original study are not universally being replicated in clinical practice. Almost half of those who tracked mortality reported seeing no benefit, and among those who did see a benefit, the majority reported mortality reductions of less than 30%, with nearly a quarter of respondents reporting a <10% reduction. Only 3% of respondents reported seeing a reduction in mortality greater than 50%. By comparison, the mortality in Dr. Marik’s initial study fell from 40% to approximately 9%, a relative reduction of 77%. So while these results may indicate some mortality reduction with use of this treatment, in actual practice, this reduction appears to be far smaller than originally reported, and likely closer to the 10-20% range if any at all. However, given the number of people affected by sepsis worldwide, even this level of reduction would be of enormous clinical benefit if subsequently validated in randomized trials, potentially leading to 10s of thousands if not 100s of thousands of lives being saved each year.
We were hoping that practitioners with higher numbers of treated patients would converge on a particular mortality reduction range, but this was not seen, perhaps due to small sample numbers. Respondents in different patient volume categories, including high volume groups, reported different mortality benefits across the spectrum. However, it did appear that those who treated more patients, and in particular >50 patients, tended to observe at least some mortality benefit vs. no benefit at all. A relative increase in the number of respondents in the 10-25 patient group was observed in the 10-30% mortality reduction range, but its significance is unclear given that many respondents in this group also reported <10% mortality benefit or no benefit. The effect of treatment timing (early vs late) on mortality was also not entirely apparent, although there did seem to be a slight benefit to earlier treatment, especially in the high >50% mortality reduction group.
Aside from lower mortality, Marik’s original study also reported a number of other positive benefits of the vitamin C cocktail including a reduction in organ dysfunction, as manifested by a lower SOFA score and lower rates of renal replacement therapy, a lower duration of vasopressor use, and a shorter length of ICU stay. Some of these effects were also reported in our survey, with many respondents reporting fewer vasopressors and a faster patient recovery, and about half this number reporting less organ dysfunction and fewer complications.
it is important to emphasize that this was an unvalidated survey whose respondents and responses were not in any way verified. The similarity of responses between the two surveys suggests that the majority of responses reflect the true experience of clinicians in the field, but these responses were largely qualitative in nature and cannot be used to draw any real conclusions about the benefits of this treatment, only speculative hints at best. We therefore do not in any way recommend or endorse use or disuse of this treatment based on the results of this survey.
We should also mention that although the ingredients of this cocktail are generally very safe in most people, there are some populations where caution should be exercised. These include people with G6PD deficiency (favism) as they may be at higher risk of hemolytic crisis with use of high dose IV vitamin C, patients with hemochromatosis as high dose vitamin C may cause abnormal iron deposition, and pregnant patients as the effects of such doses of vitamin C on developing fetuses is not known. G6PD deficiency in particular is significant because it is fairly common in many parts of the world including tropical Africa, the Middle East, subtropical Asia and parts of the Mediterranean, with a prevalence of 5-25%. In the US, approximately 10% of black males are affected by this condition. Most studies now studying this treatment list G6PD deficiency as an exclusion criterion, but its effect on this treatment is under some debate, with some arguing that it does not in fact increase the risk of hemolysis, including Dr. Marik in this recent article.
Conclusion
Our survey of 668 users of the Sepsis Clinical Guide, mostly physicians in critical care, emergency medicine and other specialties representing over 80 countries, found that 24% percent of clinicians are using the Marik vitamin C cocktail in clinical practice. Seventy-two percent of respondents reported using it in less than 25 patients, and 68% either as soon as a sepsis diagnosis is made, or only in severe sepsis and septic shock. Of those who tracked mortality, 46% reported no mortality benefit with use of the cocktail, 23% reported <10% mortality reduction, 19% reported 10-20% reduction, 7% reported 30-50% reduction and 5% reported >50% mortality reduction. Respondents with higher patient volumes tended to report higher reductions, as did those who treated earlier, but the trends were not strong and the group sample sizes too small to draw meaningful conclusions. Other beneficial effects reported by users of the cocktail included faster patient recovery and fewer vasopressors, and to a lesser degree, less organ dysfunction and fewer complications. Among those who did not use the cocktail, the most frequent reasons given for not doing so were a desire to wait for validation from larger randomized trials, and external factors like institutional prohibition on unvalidated treatments or lack of availability of the cocktail’s ingredients.
Daniel Nichita, MD

This is a valuable survey, but something was missed:
If you used the Marik protocol, did you use a restrictive fluid strategy, or did you use aggressive crystalloid resuscitation (30 ml/kg or more?) This is an important question, for large fluid volumes are dangerous. What is more, large fluid volumes are likely to wash out the concentration gradient need to drive HAT components into cells. Think about it.
Hello Matthew and thank you for your comment. We did not inquire about the fluid strategy in this survey, but do agree that large volumes of fluids can be dangerous and recommend judicious fluid use tailored to each patient’s clinical status and vitals.
As for how large fluid volumes affect HAT, these could increase the volume of distribution (Vd), especially given the leaky vasculature seen in sepsis, and therefore potentially affect dosing requirements. On the other hand, the vitamin C dose in this regimen is a mega-dose that should be more than sufficient to make up for even high levels of volume overloading. The same cannot necessarily be said for the other ingredients, which come in smaller doses. It is a good question, and we’ll look further into this and post here any answers we find.
Thanks!
Really, this is a invaluable web page.